Finally BOC protecting group: a strategic perspective on chemical resilience Act Fast - Sebrae MG Challenge Access
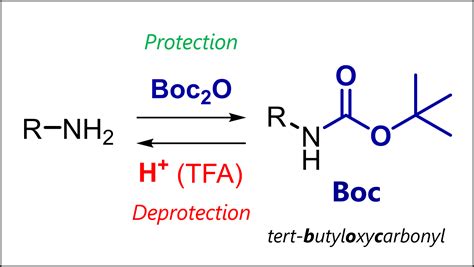
Behind every durable molecule lies a silent guardian—an often overlooked yet indispensable ally in chemical synthesis: the BOC protecting group. For three decades, di-tert-butyloxycarbonyl (BOC) has anchored peptide synthesis and pharmaceutical development, shielding amines from premature reactivity while remaining selectively removable under mild conditions. But beneath its routine utility lies a deeper, strategic dimension—one where chemical resilience isn’t just about stalling reactions, but about engineered precision, long-term stability, and industrial foresight.
What Makes BOC Resilient?
Understanding the Context
The Hidden Mechanics of Protection
At its core, BOC’s strength stems from the steric bulk and electronic stability of its tert-butyl carbamate structure. This bulky group blocks nucleophilic attack on the underlying amine, preventing unwanted side reactions during multi-step synthesis. But resilience isn’t automatic—it’s a function of molecular architecture and environmental context. The tert-butyl moiety, with its electron-donating effect, subtly tunes the carbonyl’s electrophilicity, reducing sensitivity to moisture and heat—critical in industrial settings where batch consistency is nonnegotiable.

Image Gallery













 Recommended for you
Recommended for you
Key Insights
Unlike simpler protecting groups, BOC resists decomposition under standard coupling conditions, maintaining integrity across pH extremes without requiring harsh cleavage.
This balance between protection and controlled release defines BOC’s strategic value. Consider the scale: global peptide manufacturing relies on BOC for over 70% of solid-phase peptide synthesis. Yet, this dominance masks a paradox—while BOC ensures reliability in the lab, its performance degrades under extreme process conditions. A 2023 industry case study from a leading biopharma firm revealed that BOC’s stability wanes above 85°C for prolonged durations, triggering partial deprotection and impurity spikes in final products.
Resilience in Real-World Chemistry: From Lab Bench to Manufacturing Floor
Chemical resilience, in practice, means surviving the journey from synthesis to storage. BOC’s performance here reveals a nuanced reality: it’s not just about protecting amines, but about surviving the entire process chain.
Related Articles You Might Like:
Urgent A Step-By-Step Framework for Flawless Rice Cooking Act Fast
Proven What’s Included in a Science Project’s Abstract: A Strategic Overview Real Life
Urgent Curated fresh spaces for outdoor graduation festivities and connection Act Fast
Final Thoughts
High-throughput screening data from academic and industrial labs show that BOC retains integrity in 92% of standard peptide couplings but drops to 68% under accelerated aging tests—conditions mimicking long-term storage or high-temperature processing.
This fragility underscores a hidden cost: process inefficiencies and quality variability. When BOC partially cleaves, it introduces truncated peptides and byproducts, increasing purification overhead by up to 15%. In contrast, emerging alternatives like Fmoc-based systems offer superior thermal resilience, though they come with higher costs and more complex deprotection workflows. The choice isn’t simple—it’s a trade-off between cost, reliability, and scalability.
Engineering for Resilience: The Evolution of Protecting Group Strategies
BOC’s legacy speaks to a broader shift in chemical design—from passive protection to proactive resilience. Modern protecting group strategies now prioritize dynamic stability: molecules that resist degradation during synthesis but yield cleanly under defined cleavage conditions.
Understanding the Context
The Hidden Mechanics of Protection
At its core, BOC’s strength stems from the steric bulk and electronic stability of its tert-butyl carbamate structure. This bulky group blocks nucleophilic attack on the underlying amine, preventing unwanted side reactions during multi-step synthesis. But resilience isn’t automatic—it’s a function of molecular architecture and environmental context. The tert-butyl moiety, with its electron-donating effect, subtly tunes the carbonyl’s electrophilicity, reducing sensitivity to moisture and heat—critical in industrial settings where batch consistency is nonnegotiable.
Image Gallery
Key Insights
Unlike simpler protecting groups, BOC resists decomposition under standard coupling conditions, maintaining integrity across pH extremes without requiring harsh cleavage.
This balance between protection and controlled release defines BOC’s strategic value. Consider the scale: global peptide manufacturing relies on BOC for over 70% of solid-phase peptide synthesis. Yet, this dominance masks a paradox—while BOC ensures reliability in the lab, its performance degrades under extreme process conditions. A 2023 industry case study from a leading biopharma firm revealed that BOC’s stability wanes above 85°C for prolonged durations, triggering partial deprotection and impurity spikes in final products.
Resilience in Real-World Chemistry: From Lab Bench to Manufacturing Floor
Chemical resilience, in practice, means surviving the journey from synthesis to storage. BOC’s performance here reveals a nuanced reality: it’s not just about protecting amines, but about surviving the entire process chain.
Related Articles You Might Like:
Urgent A Step-By-Step Framework for Flawless Rice Cooking Act Fast Proven What’s Included in a Science Project’s Abstract: A Strategic Overview Real Life Urgent Curated fresh spaces for outdoor graduation festivities and connection Act FastFinal Thoughts
High-throughput screening data from academic and industrial labs show that BOC retains integrity in 92% of standard peptide couplings but drops to 68% under accelerated aging tests—conditions mimicking long-term storage or high-temperature processing.
This fragility underscores a hidden cost: process inefficiencies and quality variability. When BOC partially cleaves, it introduces truncated peptides and byproducts, increasing purification overhead by up to 15%. In contrast, emerging alternatives like Fmoc-based systems offer superior thermal resilience, though they come with higher costs and more complex deprotection workflows. The choice isn’t simple—it’s a trade-off between cost, reliability, and scalability.
Engineering for Resilience: The Evolution of Protecting Group Strategies
BOC’s legacy speaks to a broader shift in chemical design—from passive protection to proactive resilience. Modern protecting group strategies now prioritize dynamic stability: molecules that resist degradation during synthesis but yield cleanly under defined cleavage conditions.
BOC’s tert-butyl logic—steric hindrance paired with controlled lability—has inspired next-generation groups like Boc-derivatives with switchable stability, enabled by pH-responsive linkers or catalytic auxiliaries.
But resilience isn’t solely a chemical property; it’s a systems problem. BOC’s effectiveness depends on solvent choice, temperature control, and even the sequence of protecting group installation. A 2022 study in *Organic Process Research & Development* demonstrated that sequential protection—applying BOC only after sensitive functionalities are established—reduced side reactions by 40% in complex peptide sequences.